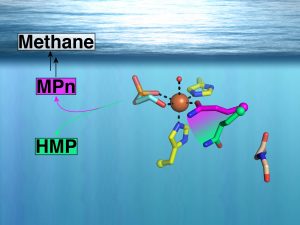
The Drennan lab began their studies of mononuclear nonheme iron enzymes with Liu lab at U Texas-Austin in the investigation of the mechanism of antibiotic fosfomycin biosynthesis by iron-dependent hydroxypropylphosphonic acid epoxidase. With the Walsh lab at Harvard Medical School, they determined the first structure of a mononuclear iron halogenase, revealing that a novel Fe metal ligation (2-His, 1-halide) is involved in the tailoring of the antifungal compound syringomycin. More recently, the Drennan lab has worked with the van der Donk lab (UIUC) to uncover sequence markers for the biosynthesis of methylphosphonate, the proposed biological source of methane from the upper ocean. The structure of methylphosphonate synthase, which they determined, led to the establishment of these sequence markers and to the discovery of another novel Fe metal ligation (2-His, 1-Gln). Importantly, these markers confirmed the abundance of methylphosphonate synthases in marine microbes, identifying a renewable source of methylphosphate, and thus methane, in the upper ocean. The newest project, a collaboration with the Zheng lab at UC Berkeley, involves the investigation of isonitrile formation by a nonheme iron enzyme. This chemistry is unprecedented in this enzyme superfamily. Overall a goal of the Drennan Lab is to provide structural insight into how natural products are biosynthesized or tailored by a family of metalloenzymes with this relatively simple cofactor, a single iron bound to two or three protein side chains. Thus far the structures have been full of surprises.
Press
MIT News, the MIT Energy Initiative News, and by the Argonne National Laboratory (2017): “Researchers Establish Long-Sought Source of Ocean Methane” by Anne Trafton
Nature (2013): “Biochemistry: Positive and radical” by Spencer C. Peck & Wilfred. A. van der Donk, vol. 496, 34-35.
Publications(‡Co-first authors, *Corresponding authors)
Adak‡, S., Ye‡, N., Calderone, L.A., Duan, M., Lubeck, W., Schäfer, R.J.B., Lukowski, A.L., Houk, K.N., Pandelia*, M.-E., Drennan*, C.L., Moore, B.S. (2024) Oxidative rearrangement of tryptophan to indole nitrile by a single diiron enzyme, Nat. Chem. doi:10.1038/s41557-024-01603-z. PMID: 37577561
Jonnalagadda‡, R., Del Rio Flores‡, A., Cai, W., Mehmood, R., Narayanamoorthy, M., Ren, C., Zaragoza, J.P.T., Kulik*, H.J., Zhang*, W., and Drennan*, C.L. (2021) Biochemical and Crystallographic Investigations into Isonitrile Formation by a Nonheme Iron-Dependent Oxidase/Decarboxylase, J. Biol. Chem. 296, 100231. doi: 10.1074/jbc.RA120.015932. PMCID: PMC7949033. PMID: 33361191
Harris, N.C., Born, D.A., Cai, W., Huang, Y., Martin, J., Khalaf, R., Drennan, C.L., and Zhang*, W. (2018) Isonitrile Formation by a Non-Heme Iron(II)-Dependent Oxidase/Decarboxylase, Angew. Chemie Int. Ed. 57(31), 9707-9710. doi: 10.1002/anie.201804307. PMCID: PMC6191297. PMID: 29906336
Born‡, D.A., Ulrich‡, E.C., Ju, K.-S., Peck, S.C., van der Donk*, W.A., and Drennan*, C.L. (2017) Structural Basis for Methylphosphonate Biosynthesis, Science 358, 1336-1339. PMCID: PMC5901744
Chang, W-C., Dey, M., Liu, P., Mansoorabadi, S.O., Moon, S-J, Z.K., Drennan, C.L., and Liu*, H-W. (2013) Mechanistic Studies of an Unprecedented Enzyme-catalyzed 1,2-Phosphono Migration Reaction, Nature 496, 114-118. PMCID: PMC3725809
Kulik*, H.J., and Drennan, C.L. (2013) Substrate Placement Influences Reactivity in Non-heme Fe(II) Halogenases and Hydroxylases, J. Biol. Chem. 288, 11233-11241. PMCID: PMC3630895
Kulik*, H.J., Blasiak, L.C., Marzari, N., and Drennan, C.L. (2009) First Principles Study of Non-heme Fe(II) Halogenase SyrB2 Reactivity, J. Am. Chem. Soc. 131, 14426–14433. PMCID: PMC2760000
Yun, D., Dey, M., Higgins, L.J., Yan, F., Liu, H.-w., and Drennan*, C.L. (2011) Structural Basis of Regiospecificity of a Mononuclear Iron Enzyme in Antibiotic Fosfomycin Biosynthesis, J. Am. Chem. Soc. 133, 11262–11269. PMCID: PMC3140168
Wong, C., Fujimori, D.G., Walsh, C.T., and Drennan*, C.L. (2009) Structural Analysis of an Open Active Site Conformation of Nonheme Iron Halogenase CytC3, J. Am. Chem. Soc. 131, 4872-4879. PMCID: PMC2663892
Blasiak, L.C., Vaillancourt, F.H., Walsh, C.T., and Drennan*, C.L. (2006) Crystal Structure of the Non-haem Iron Halogenase SyrB2 in Syringomycin Biosynthesis, Nature 440, 368-371. Faculty of 1000 Recommended
Higgins, L. J., Yan, F., Liu P., Liu, H.-W., and Drennan*, C. L. (2005) Structural Insight into Antibiotic Fosfomycin Biosynthesis by a Mononuclear Iron Enzyme, Nature 437, 838–844. Faculty of 1000 Recommended